
Reduce reviewer-to-reviewer variability and minimize missed findings. It also automatically checks consistency with prior review comments and source references.
*1 Experienced professionals with backgrounds in pharmaceutical material creation, review operations, and drug discovery support.

REVIEW TEAMS

MARKETING TEAMS

AI technology plus expert review support makes fast, high-quality review possible.
Register the materials to be reviewed together with the documents used as review criteria, such as package inserts, interview forms, CTD files, company policies, and slide kits.
The review AI can analyze tables, graphs, and images as well as text. A second check by experienced specialists*1 helps maintain review quality.
Findings are delivered together with their supporting rationale, making downstream revision and confirmation work easier to manage.
Lecture slides
Review comments
*1 Experienced professionals with backgrounds in pharmaceutical material creation, review operations, and drug discovery support.
Compared with conventional outsourced review, Shaperon delivers faster turnaround.
Expert double-checks*1 also help prevent critical misses and mistakes.
3 to 5 business days

Within 2 business days *2
*1 Experienced professionals with backgrounds in pharmaceutical material creation, review operations, and drug discovery support.
*2 For standard lecture slides. Actual processing time varies depending on the number of pages and the types of materials uploaded.
Reduce the burden on review and medical teams while improving both quality and efficiency.
The standard review algorithm is built around promotional activity guidelines and content production standards, and can flexibly adapt to each company’s policies and proprietary rules.
The AI can flexibly manage company-specific review criteria and scope, including how competitor brand names are handled and whether trial comparisons are permitted. Multiple standards can be maintained at the same time.
| Company A | Company B | |
|---|---|---|
| Competitor brand names | Flag Generic name alongside is also not allowed | No flag needed |
| Comparisons across different trials | Flag All comparisons are not allowed | Flag Comparisons with competitor products are not allowed |
Automatically compare the materials under review with past slides by the same speaker. This helps maintain consistency with prior comments and prevents missed findings or wording drift.

Lecture slides
under review

Past slide review results
for the same speaker
Automatically compare graphs and numeric claims in slides against public literature sources such as PubMed. The system detects inconsistencies with the source and exaggerated expressions.
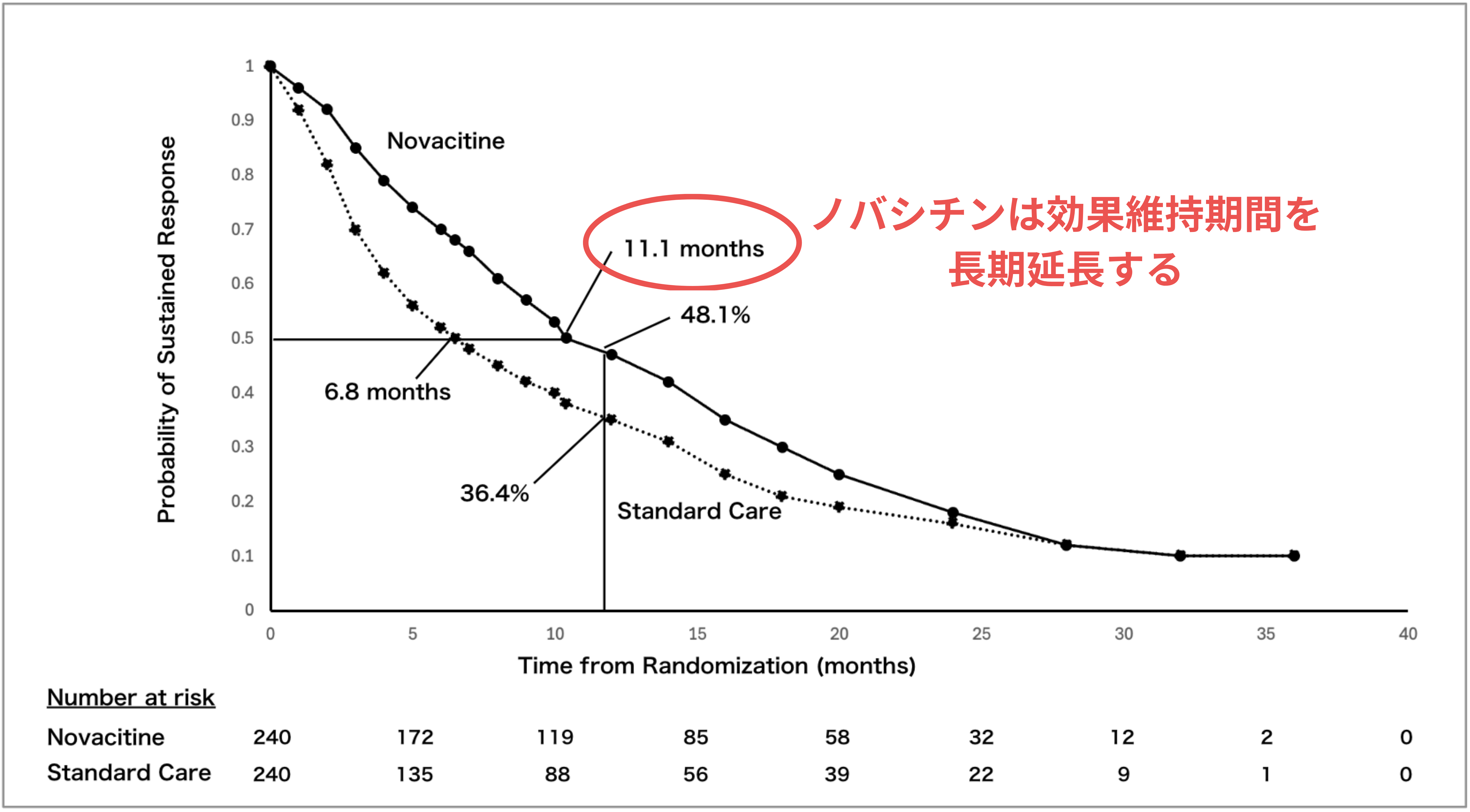
Lecture slide
before review
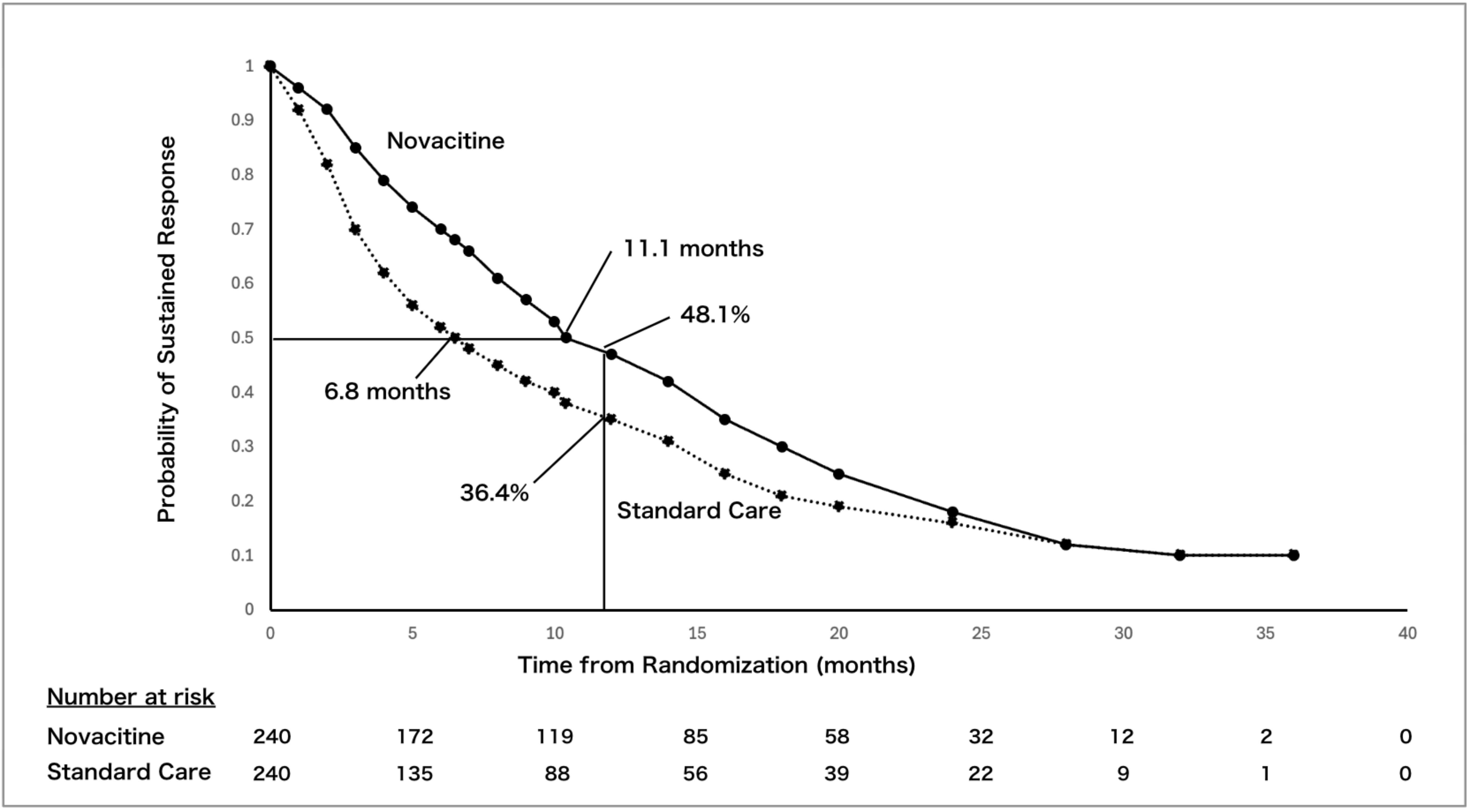
Public reference data
such as PubMed
*3 Based on lecture-slide review after stable production deployment. Actual results vary by use case and operating conditions.
Contact
Start by contacting us. We can discuss the materials you want to review and answer any questions you have about the service.
Scope and estimate
We review material types, product count, monthly volume, desired rollout timing, and any other requirements, then provide a feasibility assessment and estimate.
Full implementation
Before launch, we align with your review standards and operational workflow. After implementation, we continue improving performance to support your review operations over time.
Beyond the specific use cases shown on this page,
we can also discuss broader review and compliance challenges unique to the pharmaceutical industry.